DARA BioSciences, Inc. (DARA) stock posted today 20.95% in gains on 6,424,094 shares in trading volume and brought to followers for about 55% in profits since our alert issued on September 13th.
Our “gut feeling” and quick observation of DARA’s financials “told” us that a second offering for this biotech penny markets most likely is on the way… To make a long story short – we took a profit.
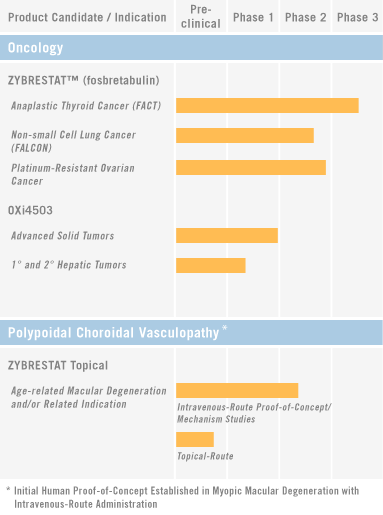
On the other hand, OXiGENE, Inc. (OXGN) announced today that it has received SPA (Special Protocol Assessment) for its Phase 3 clinical trial (FACT 2) of ZYBRESTAT for the treatment of anaplastic thyroid cancer (ATC) from U.S. Food and Drug Administration (FDA).
The FACT 2 study is designed as a randomized, placebo-controlled, double-blinded study of 300 subjects randomized 1:1 to receive carboplatin and paclitaxel plus fosbretabulin versus carboplatin and paclitaxel plus placebo. The primary endpoint of the study will be to compare the median overall survival between the two arms.
We joined OXGN at $.68/share and alerted our Twitter and Facebook followers. Bottom line is even OXiGENE’s ATC drug wouldn’t be the major company’s revenue maker (when approved), but it could be a candidate for the FDA’s fast-track. And there is why:
Anaplastic thyroid cancer invades adjacent structures and metastasize extensively to cervical lymph nodes and distant organs, such as the lungs and bones. Tracheal invasion is present in 25% at the time of presentation (said differently, in about 25% of cases, the anaplastic cancer has grown out of the thyroid and into the trachea). This is why many patients with anaplastic thyroid cancer will need a tracheostomy, while almost nobody with the other types of thyroid cancer will need one.
Anaplastic thyroid cancer that spreads to the lungs is present in 50% of patients at the time of diagnosis. Most of these cancers are so aggressively attached to vital neck structures that they are inoperable at the time of diagnosis. (The surgeon can’t remove it because it’s growing into other neck structures). Even with aggressive therapy protocols, such as hyperfractionated radiation therapy, chemotherapy, and surgery, survival at 3 years is less than 10%.
Significant increase of the overall survival rate of ZYBRESTAT’s patients is a straight path to the fast-track FDA approval.
Peter J. Langecker, M.D., Ph.D., OXiGENE’s President and Chief Executive Officer, commented in today’s press release:
…We are hopeful that the receipt of the SPA will strengthen our ability to obtain funding to conduct the FACT 2 study, either through financing or partnering. We believe that vascular disrupting agents (editor note: read “Tumor Vasculature: a Target for Anticancer Therapies” by Dr. Dietmar W. Siemann) can make a valuable contribution to anticancer therapeutic regimens, and we look forward to advancing our VDA programs not only in ATC, but also in ovarian cancer, carcinoid syndrome, myeloid leukemia and other potential indications.
 OXiGENE, Inc.
OXiGENE, Inc.
701 Gateway Blvd, Suite 210
South San Francisco, CA 94080
Tel: (650) 635-7000
Fax: (650) 635-7001
