Alzheimer’s disease, the most common form of dementia, is characterized by progressive loss of memory and cognition, ultimately leading to complete debilitation and death. Currently marketed drugs transiently affect some symptoms of the disease, but there are no drugs on the market today that slow or arrest the progression of the disease. Intellect Neurosciences, Inc. is a Manhattan -based biopharmaceutical company engaged in the discovery and development of disease-modifying therapeutic agents for the treatment and prevention of Alzheimer’s disease and other disorders. These symptomatic drugs are projected to generate more than US $6 billion in sales this year, indicating both the size of the market and the demand for effective treatment beyond symptomatic improvements.
Intellect Neurosciences Inc. (OTCBB: ILNS) is a biopharmaceutical company engaged in the discovery and development of a new breed of innovative “disease-modifying” therapeutic drugs designed to slow, arrest and ultimately prevent Alzheimer’s disease and other serious aging disorders.
45 West 36th Street, 3rd Floor
New York, NY 10018
Tel: 212 448 9300
Fax: 212 448 9600
http://www.intellectns.com
Contact:
Elliot Maza , JD, CPA, President and Chief Financial Officer
Tel: 212-448-9300
Daniel G. Chain, Ph.D.
Chief Executive Officer and Chairman
Dr. Chain formed Intellect in April 2005 and has served as our Chairman and Chief Executive Officer since October 2005. Dr. Chain was previously the President and CEO of Mindset BioPharmaceuticals, a private company he founded in 1999.
Dr. Chain is the inventor of the Company’s ANTISENILIN® technology for the treatment and prevention of Alzheimer’s disease which underlies products in Phase 1, Phase 2 and Phase 3 clinical trials by major pharmaceutical companies that have purchased licenses from Intellect Neurosciences for the commercialization of those products. Patents in relation to Dr. Chain’s invention have been granted in Europe, Japan and several other countries and are pending in the United States. Dr. Chain is also the inventor of a patent recently granted in Europe concerning the use of insulin sensitizers for the prevention and treatment of memory loss and dementia. Dr. Chain has spend more than a decade devoted to developing innovative approaches aimed at slowing, arresting or preventing Alzheimer’s disease. Collectively, these different approaches drive Intellect Neuroscience’s internal drug pipeline that is backed by intellectual property assets across diversified technologies. Dr. Chain has authored numerous scientific research publications in peer-reviewed journals and his broad expertise is increasingly drawing positive attention in the media featuring in articles and radio interviews to discuss new developments in the Alzheimer’s field and the progress being made by Intellect Neurosciences. Dr. Chain is a member of the Royal Society of Medicine and the Society for Neuroscience. Dr. Chain obtained his B.Sc., with honors, in Biochemistry from the Institute of Biology in London and obtained his Ph.D. in Biochemistry from the Weizmann Institute of Science in Israel. He trained as a post-doctoral research fellow at the Center for Neurobiology and Behavior at Columbia University College of Physicians and Surgeons in New York where he collaborated with Nobel Laureate, Professor Eric Kandel.
Elliot M. Maza, J.D., CPA
President and Chief Financial Officer
Mr. Maza has served as our President and Chief Financial Officer since March 2007. He served as Executive Vice President and Chief Financial Officer from May 2006 until March 2007. From December 2003 to May 2006 Mr. Maza was Chief Financial Officer of Emisphere Technologies, Inc., a publicly held biopharmaceutical company specializing in oral drug delivery. He was a partner at Ernst and Young LLP from March 1999 to December 2003 and an investment banker at Goldman Sachs, Inc., JP Morgan Securities, Inc. and Bankers Trust Securities, Inc. at various times during April 1989 to March 1999. Mr. Maza practiced law at Sullivan and Cromwell in New York from September 1985 to April 1989. Mr. Maza serves on the Board of Directors and is Chairman of the Audit Committee of Apollo Solar Energy, Inc., a Chinese producer of material used to manufacture solar cells. Mr. Maza received his B.A. degree from Touro College in New York and his J.D. degree from the University of Pennsylvania Law School. He is a licensed C.P.A. in the states of New Jersey and New York and is a member of the New York State Bar.
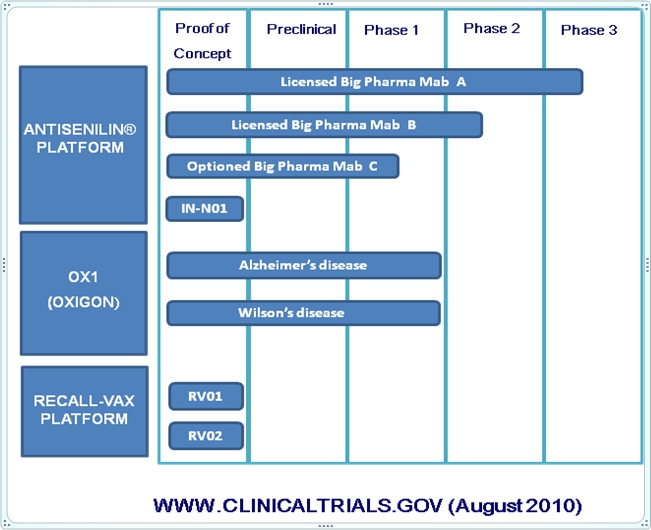
Platform Technologies:
The most advanced internal product in Intellect Neurosciences’ drug development pipeline is OXIGON™ (OX1). OX1 is an orally-administered small molecular weight copper-binding drug compound that prevents oxidative stress and blocks formation of toxic Aß aggregates and is being developed by the Company as a potential disease-modifying treatment for AD. In addition, the Company hopes to establish proof of concept for other diseases, including Wilson’s disease (“WD”), a hereditary Orphan Disease indication affecting about 1 in 30,000 people that has multiple neurological and other symptoms caused by copper overload and oxidative stress. The development of ILNS-OX1 has received financial support from the Institute for the Study of Aging, the BIRD Foundation and the National Institute of Aging, part of the National Institutes of Health. The Company plans to conduct a Phase II biomarker trial for OX1 in AD patients. The objective of the trial is to test safety in AD patients and demonstrate potential changes in relevant biomarkers.
In addition, the company has two preclinical programs it is developing internally. One of these, under the ANTISENILIN platform, is IN-N01, which the Company is developing as a follow-on generation monoclonal antibody drug that it believes will have better safety and efficacy in a broader population than Bapineuzumab or Ponezumab and compared to other similar types of drugs currently in development. The resulting product has multiple potential applications for the treatment of AD, Glaucoma, Age-related Macular Degeneration and Traumatic Brain Injury, all of which have been linked to A?. The second program under the RECALL-VAX platform has yielded two AD vaccine drug candidates, RV01 and RV02. RECALL-VAX™ has the potential to delay or prevent the development of Alzheimer’s in those who are at risk by stimulating patients’ immune systems to produce antibodies that clear amyloid betas and prevent further accumulation and deposition. Many view a vaccine of this nature, which is analogous to a flu shot, as the ultimate quest in Alzheimer’s research.
Intellect Neurosciences, Inc. Obtains Pharmacokinetic Profile Report From Fourteen Day Safety Trial for the Company’s Lead Alzheimer’s Candidate, OX1
NEW YORK , Dec. 27, 2010 (GLOBE NEWSWIRE) — Intellect Neurosciences, Inc. (OTCBB:ILNS), a biopharmaceutical company with an internal preclinical and clinical-stage pipeline and multiple licenses with major pharmaceutical companies covering products in late-stage clinical trials, announced that it has obtained a draft pharmacokinetic report from its completed Phase 1b trial for its lead Alzheimer’s candidate, OX1. The Company previously reported on the safety aspects of the trial, which demonstrated that OX1 is safe and well tolerated at all dose levels tested.
The pharmacokinetic data indicates that the drug is rapidly absorbed and distributed in the body after oral administration. Moreover, the terminal elimination half-life increased from approximately 6-8 hours on the first day of the trial to 8-9 hours on the fourteenth day.
The draft report was provided by Kendle , a global clinical research organization, which conducted the Phase 1b trial through the Kendle Clinical Pharmacology Unit located in Utrecht , The Netherlands.Intellect is the sponsor of the trial, which consisted of 14 days of repeated dosing in a double blind, randomized, placebo-controlled, multiple escalating dose study with 3 groups of 12 healthy elderly volunteers aged 60 or more.Subjects in each group, consisting of 4 subjects receiving placebo and 8 subjects receiving OX1, were administered daily doses of 200, 400 or 800 mg at quarterly (6 hour) intervals.
Dr. Daniel Chain , Chairman and Chief Executive Officer of Intellect, commented: “The increase in terminal elimination half-life suggests that the drug could be administered less frequently than in our completed Phase 1 trials. We are pleased to receive this data, which we will use in designing our planned Phase 2 trial, where we aim to demonstrate the drug’s effect in Alzheimer’s patients on a relevant biomarker in the cerebrospinal fluid.”
In a separate development, the Company announced that the National Institute of Aging (NIA), part of the National Institutes of Health , will support toxicology studies for OX1 as part of a federal effort to work with academia and the private sector to encourage the discovery and development of drugs for Alzheimer’s disease.The NIA previously supported key safety and toxicology studies for OX1, which were conducted by the Biosciences Division of SRI International , Menlo Park , California.Dr. Chain commented: “We are delighted that the NIA will support additional development work needed for future clinical studies of OX1, which we expect to undertake early in the New Year.”
Dr. Chain continued: “OX1 is a small molecule that works through a completely different mechanism of action than our products that are focused on immunotherapy-based approaches. OX1 not only promotes clearance of soluble beta amyloid but also directly blocks neurotoxicity caused by free radical generating reactions that damage synaptic function in the brain and ultimately give rise to irreversible changes inside nerve cells.We have received interest from several large pharmaceutical companies regarding OX1 and anticipate partnering this drug in the future. We look forward to moving forward with our clinical and regulatory development plan that includes the filing of an Investigational New Drug application (IND) with the FDA to support initiation of Phase 2 trials. Typically, licenses and or collaborations with large pharmaceutical companies yield substantial revenues from license fees, development milestone payments and royalties from sales.”