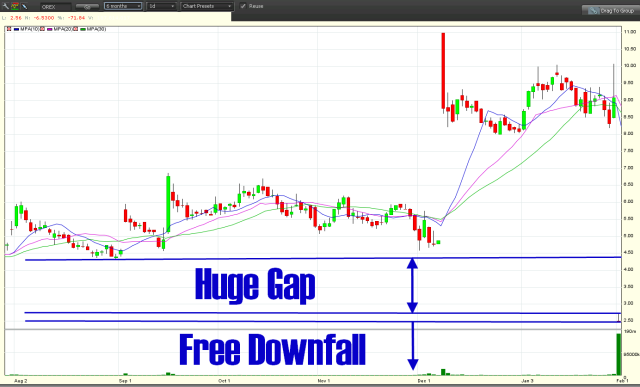
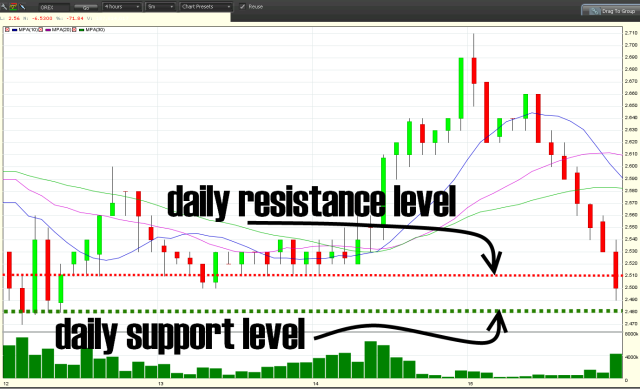
Shares of Orexigen Therapeutics (OREX) plunged Tuesday after the Food & Drug Administration denied approval of its application for a new obesity drug.
But does it justify a 72% down day on volume of 4X the company’s issued & outstanding (o/s) share count?
Orexigen had developed, Contrave, a weight-loss drug, in partnership with Japan ‘s Takeda Pharmaceutical Company.
Orexigen said the FDA sent it a complete response letter outlining concerns about the long-term cardiovascular safety of the drug. The FDA said that in order for the application to be approved, the company must conduct a randomized, double-blind, placebo-controlled trial of a sufficient size and duration to demonstrate the potential cardiovascular risk to analyze whether it outweighs the benefits of the drug.
Orexigen said it will work with the FDA to determine its next steps.
Back in December, an FDA panel had voted to recommend approval of Contrave, but by a slim margin, and the agency is not required to accept the recommendations of its committees.
The FDA rejected obesity drugs from Arena Pharmaceuticals (ARNA) and Vivus (VVUS) over concerns for the potential risks of the drugs back in October.
Since then, VVUS ran from $5 to $11; ARNA jumped from $1.30 to $2.70. ‘Nuff said.
6 month & daily charts: