By Mr. BiotechIll, Guest Author
Shares of Peregrine Pharmaceuticals Inc. (NASDAQ: PPHM) gain almost half their value after announcement that interim results were presented from its 121 patient randomized, double-blind, placebo-controlled Phase IIb trial in patients with refractory non-small cell lung cancer (NSCLC).
At glance:
- Data show statistically significant improvement in overall survival for patients receiving Bavituximab plus Docetaxel versus Docetaxel alone
- Clinical data strongly support advancing program into Phase III clinical development
Steven W. King, president and chief executive officer of Peregrine, stated:
Robust data from this Phase II trial clearly demonstrate a significant benefit in overall survival with a good safety profile in patients receiving bavituximab plus docetaxel compared to those receiving docetaxel plus placebo. We are currently in discussions with several potential pharmaceutical partners who have expressed great interest in our bavituximab oncology program. It is our goal to identify the optimal partner to assist with the design and logistics of a multinational Phase III pivotal trial
Company to host conference call on Monday, September 10, at 11:00 AM EDT: please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals call.
Please Note:
- Final results from Bavituximab’s trial are far from assured
- Overall results will not be available until the end of this year
- Phase III pivotal trial planned to be only in the second quarter of next year
- Only $18,033,000 cash in hands vs. $35,688,000 R&D expenses in fiscal year 2012
Summary:
- Second offering is on the way if Peregrine wouldn’t be able to find partner for the launch of Bavituximab’s trial
- PPHM stock is heavily overpriced
- Share/price correction is coming.
Peregrine Pharmaceuticals’s pipeline:
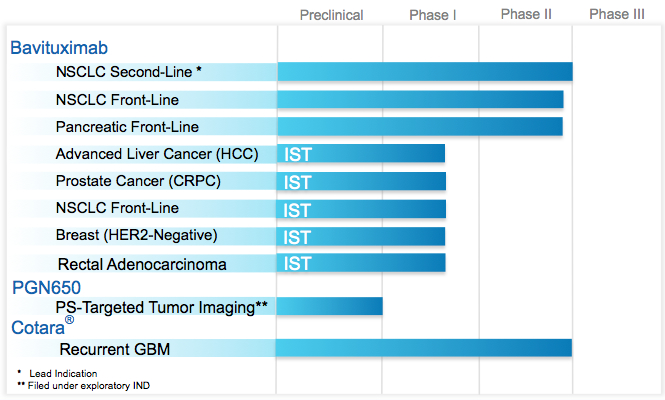
Bavituximab Oncology
Currently in Phase II clinical development, bavituximab is a first-in-class phosphatidylserine (PS)-targeting monoclonal antibody that has demonstrated broad therapeutic potential across multiple oncology indications and represents a new approach to treating cancer. Bavituximab is currently being evaluated in three randomized Phase II clinical trials for non-small cell lung cancer (NSCLC) and pancreatic cancer.
Cotara Oncology
The lead candidate from our Tumor Necrosis Therapy (TNT) platform, Cotara is a novel therapy in Phase II clinical development for the treatment of glioblastoma multiforme (GBM), the deadliest form of brain cancer. Cotara links a radioactive isotope to a targeted monoclonal antibody designed to bind to the DNA histone complex that is exposed by dead and dying cells found at the center of solid tumors. Cotara’s targeting mechanism enables it to bind to the dying tumor cells, delivering its radioactive payload to the adjacent living tumor cells and essentially destroying the tumor from the inside out, with minimal radiation exposure to healthy tissue. Cotara is delivered in a single dose using convection-enhanced delivery (CED), an NIH-developed method that targets the specific tumor site in the brain.
 Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc.
14282 Franklin Avenue,
Tustin, CA 92780
Tel: 714-508-6000
Fax: 714-838-5817
Peregrine Pharmaceuticals (Nasdaq: PPHM) is a clinical-stage biopharmaceutical company developing and manufacturing innovative monoclonal antibody therapeutics for the treatment of cancer and viral infections.
